GLP-1 medications are reshaping metabolic care, but the path from prescription to staying on therapy is rarely straightforward. Coverage, prior authorization, side effects, dose escalation, and long-term planning are separate problems, and each one can stop someone from continuing a medication that is working. This page collects every tool and guide we have built for patients on GLP-1 therapy, so you do not have to hunt for them.
Before you start: important safety information.
GLP-1 receptor agonists (semaglutide, tirzepatide, and others in this class) carry a boxed warning from the U.S. Food and Drug Administration for the risk of thyroid C-cell tumors, based on findings in rodents. The clinical relevance in humans is not fully established, but the warning is on the label of every product in this class.
These medications should not be used in people with:
- A personal or family history of medullary thyroid carcinoma (MTC)
- Multiple Endocrine Neoplasia syndrome type 2 (MEN 2)
- A history of serious hypersensitivity to the specific agent
Additional label warnings to discuss with your clinician include the risk of acute pancreatitis, gallbladder disease, acute kidney injury (often related to dehydration from nausea or vomiting), and for some agents, diabetic retinopathy complications in patients with type 2 diabetes. GLP-1 therapy should be discontinued in pregnancy. None of this is a substitute for your prescribing clinician's judgment about whether a GLP-1 medication is appropriate for you, and it is not a reason to stop therapy on your own without that discussion.
Source: FDA prescribing information for Wegovy (semaglutide) and Zepbound (tirzepatide). Always review the current label for the specific product prescribed.
Use our GLP-1 tools
Two tools on AnchorWellPress are built specifically for GLP-1 therapy.
GLP-1 Side Effect Coach
Personalized guidance for managing GLP-1 side effects, dose-escalation timing, and nutrition strategy between appointments. Flags clear signals for when a symptom is worth a call to your clinician.
Open the Side Effect Coach →GLP-1 Insurance Navigator
A coverage walkthrough for the most common GLP-1 medications (Ozempic, Wegovy, Mounjaro, Zepbound). Explains why prior authorization is so often required and what an approval pathway looks like on your plan type.
Open the Insurance Navigator →Not sure where to start?
If the question in front of you is about cost, coverage, or a prior authorization, begin with the Insurance Navigator. If you are already on a GLP-1 and managing symptoms, start with the Side Effect Coach.
If prior authorization is your hurdle
Most GLP-1 coverage challenges are a documentation problem, not a clinical one. These AnchorWellPress tools support the full prior authorization and appeal journey.
Benefits Quiz
A two-minute walkthrough that tells you what kind of plan you have (commercial, Medicare, Medicaid, self-funded) and what that means for GLP-1 coverage pathways. Start here if you are not sure what your plan actually does.
Take the quiz →PA Criteria Reference
A plain-language reference for the clinical criteria most plans use to evaluate GLP-1 prior authorization. Know what your plan is looking for before the request is submitted.
Open the reference →Prior Authorization Toolkit
A step-by-step guide for patients and clinical staff preparing a GLP-1 prior authorization request, including documentation checklists and common denial reasons.
Open the toolkit →Medical Necessity Letter Generator
A structured letter of medical necessity your prescribing clinician can adapt and sign, written to match the coverage criteria most insurers apply to GLP-1 therapy.
Generate a letter →Patient Appeal Generator
If your GLP-1 prior authorization was denied, this tool helps you draft an appeal that addresses the plan's specific denial language and requests the right level of review.
Draft an appeal →Read the guide
An evidence-based patient guide on GLP-1 therapy, written for the question most people actually ask: what is happening in my body, and what does it mean for the next year?
Patient Guide
Understanding Weight Changes After GLP-1 Therapy: What the Research Shows — how GLP-1 medications actually work in the brain and gut, what clinical trials show about weight loss and regain, and what the research means for long-term planning. Includes an embedded video companion.
Companion product
A paperback tracker designed to sit on a kitchen counter or nightstand during the first months of therapy.
Paperback · Available on Amazon
GLP-1 Companion Journal & Tracker — an evidence-based paperback tracker for medication, nutrition, body composition, and side effects during semaglutide or tirzepatide therapy.
Watch & learn
Three short explainers from the AnchorWellPress YouTube channel. Each is six minutes or less and pairs with the written guide above.
What Happens After Ozempic? Understanding Weight Rebound After GLP-1 Therapy — why weight returns for most patients who stop GLP-1 therapy, and what the STEP and SURMOUNT trial extensions actually show. (6:08) The GLP-1 Muscle Tax: What Are You Really Losing? — weight lost on a GLP-1 includes lean mass, not just fat, and what that means for protein, training, and long-term function. (5:36) Should You Start Training Before Starting a GLP-1? — the case for a brief resistance-training base before initiating therapy, and what the early weeks look like with and without it. (6:00)More short videos on muscle preservation, metabolic adaptation, and GLP-1 strategy on the AnchorWellPress YouTube channel.
What the research shows
Three findings that shape how we talk about GLP-1 therapy on this platform.
 Three findings at a glance — full citations below.
Three findings at a glance — full citations below.
Muscle preservation matters
Weight lost on GLP-1 therapy includes both fat and lean mass. Protein intake at roughly 1.2 to 1.6 grams per kilogram of body weight, combined with resistance training two to three times per week, is associated with preserving more lean mass during weight loss in published trials.
Adapted from current obesity medicine guidance; discuss specifics with your clinician.
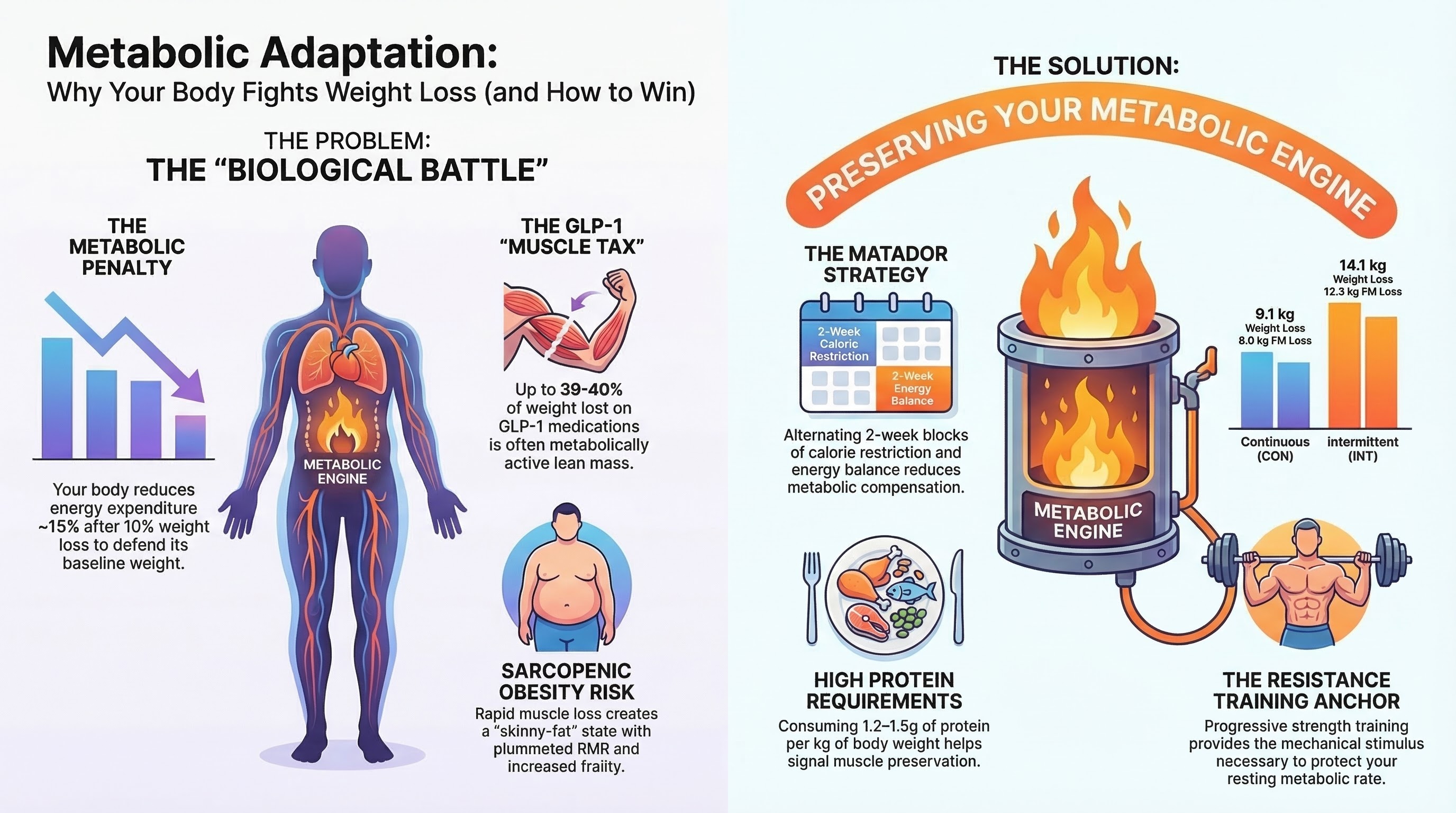 Metabolic adaptation and the muscle tax during GLP-1 therapy.
Metabolic adaptation and the muscle tax during GLP-1 therapy.
Weight regain after stopping is the norm, not the exception
In the STEP 1 extension trial, patients who stopped semaglutide regained roughly two-thirds of the weight they had lost within one year. In the SURMOUNT-4 trial of tirzepatide, patients who switched to placebo regained about half of the weight they had lost over 52 weeks. This is a medication-class pattern, not a personal failure. It informs decisions about how long to stay on therapy, and whether dose tapering is appropriate.
Wilding JPH et al., Diabetes Obes Metab 2022; Aronne LJ et al., JAMA 2024.
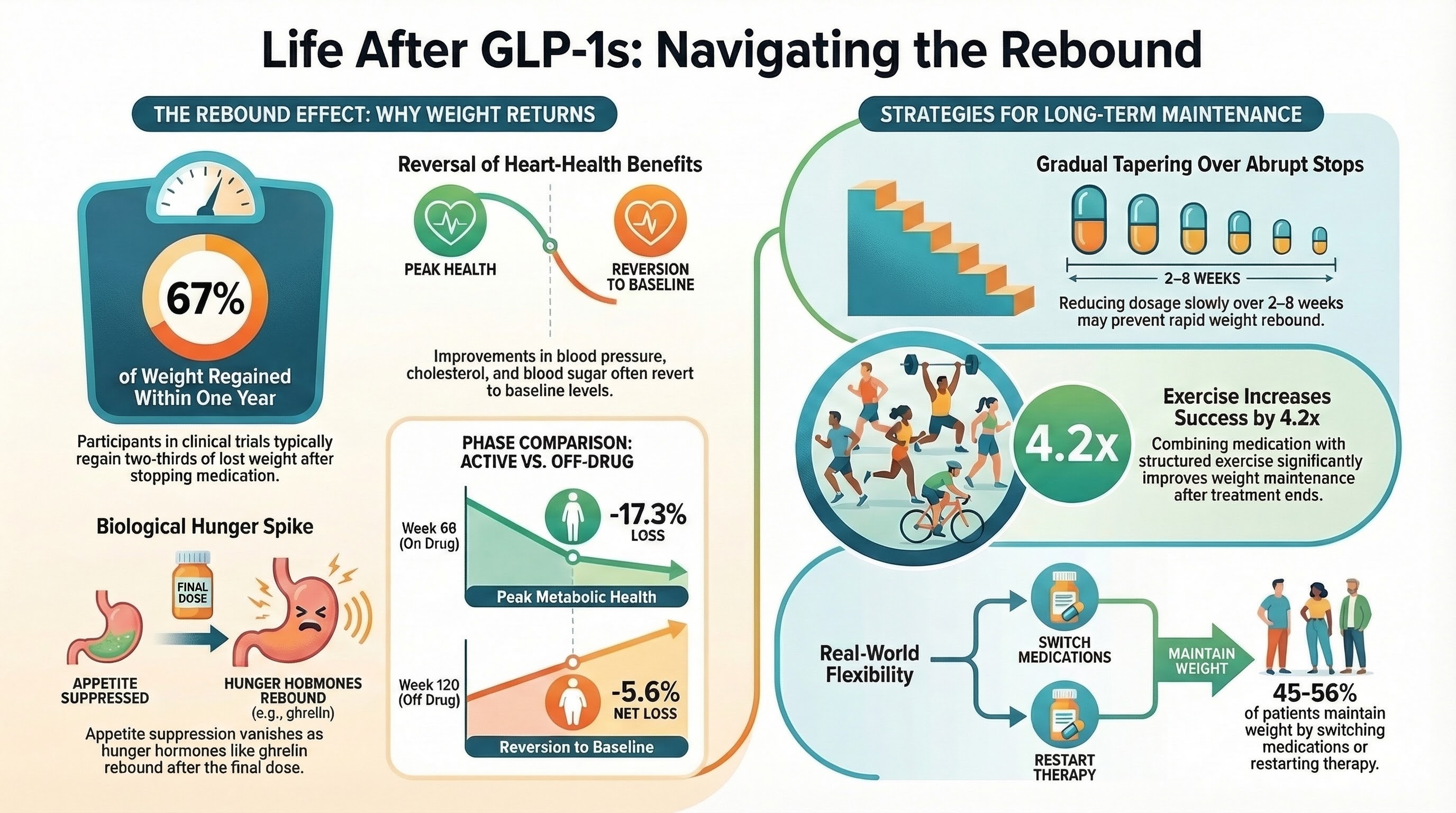 What happens biologically after stopping a GLP-1, and what improves long-term maintenance.
What happens biologically after stopping a GLP-1, and what improves long-term maintenance.
Prior authorization approval depends on documentation, not persuasion
Insurers evaluate prior authorization requests against written clinical criteria. The strongest predictor of approval is whether the submitted documentation maps to those criteria: qualifying diagnosis, required labs where applicable, documented prior treatments where required, and correct dosing. A well-prepared request is approved more often than one that is not.
Synthesized from published payer criteria; exact requirements vary by plan.
Frequently Asked Questions
Four GLP-1 receptor agonists are most commonly prescribed in the United States. Ozempic (semaglutide) and Mounjaro (tirzepatide) are FDA-approved for type 2 diabetes. Wegovy (semaglutide) and Zepbound (tirzepatide) are FDA-approved for chronic weight management in adults who meet specific BMI and comorbidity criteria. Wegovy also has an FDA-approved cardiovascular risk-reduction indication for adults with established cardiovascular disease and overweight or obesity, and Zepbound received an expanded FDA indication in December 2024 for moderate-to-severe obstructive sleep apnea in adults with obesity. The active ingredient is the same across indications for each molecule; the dose, labeling, and insurance coverage pathway are what differ.
Prior authorization is an insurer's way of confirming, before it pays, that a medication meets the plan's clinical coverage criteria. For GLP-1s, the criteria typically include a qualifying diagnosis (type 2 diabetes, or obesity with defined BMI and comorbidities), documented prior treatment attempts where required, and sometimes lab values like A1C. The list price of these medications is high, so insurers apply utilization management. Prior authorization is not the same as a denial. It is a documentation step, and a well-prepared request is approved more often than one that is not.
Most patients regain a meaningful share of lost weight after stopping a GLP-1, because the medication works on appetite regulation and that effect ends when the drug clears. The exact pattern differs by drug. In the STEP 1 extension trial of semaglutide, patients regained about two-thirds of the weight they had lost within one year of stopping. In the SURMOUNT-4 trial of tirzepatide, patients who switched to placebo regained about half of the weight they had lost over 52 weeks, and roughly four in five regained at least a quarter of their peak weight loss. Regain after any weight intervention is a common pattern in obesity medicine and does not reflect personal failure. How long to stay on therapy, and whether dose tapering is appropriate, are decisions to make with your prescribing clinician.
The most common side effects, especially in the first weeks after a dose increase, are nausea, reduced appetite, constipation, and reflux. Most resolve as the body adapts. Helpful strategies include eating smaller meals more slowly, prioritizing protein and hydration, avoiding high-fat or large meals at the start of dose escalation, and keeping a symptom log to share with your clinician. Persistent vomiting, severe abdominal pain, or signs of dehydration should prompt a call to your prescriber rather than self-management.
Semaglutide (Ozempic, Wegovy) is a GLP-1 receptor agonist. Tirzepatide (Mounjaro, Zepbound) is a dual agonist that activates both GLP-1 and GIP receptors. In head-to-head and comparative studies, tirzepatide has generally produced greater average weight reduction, but both medications work, both require dose escalation to manage side effects, and both involve similar insurance considerations. The right choice depends on your clinical picture, insurance formulary, and tolerability.